
This would mean that indium’s atomic mass was actually 113, placing the element between two other metals, cadmium and tin. An atomic dining table can differ in price owing to various characteristics the average selling price 1stDibs is 1,275, while the lowest priced sells for 235 and the highest can go for as much as 12,328. Because elemental indium is a silvery-white metal, however, Mendeleev postulated that the stoichiometry of its oxide was really In 2O 3 rather than InO. If this atomic mass were correct, then indium would have to be placed in the middle of the nonmetals, between arsenic (atomic mass 75) and selenium (atomic mass 78). Pd, having the closed-shell 4d 10 5s 0 electron configuration, was not included in the fit for the 4d elements (Figure 3(b)). Visualize trends, 3D orbitals, isotopes, and mix compounds. Plot of atomic polarizabilities as a function of atomic number for the d-block and f-block elements together with weighted, nonlinear least-squares fits according to empirical power relationships between and Z. The atomic mass of indium had originally been reported as 75.6, based on an assumed stoichiometry of InO for its oxide. Interactive periodic table showing names, electrons, and oxidation states. Atomic Actinoids (Actinides) Lanthanoids (Lanthanides) Noble gases Other nonmetals Post-transition. Since we are adding electrons to the 3d orbitals, we are actually adding to the core electrons and not to the valence orbitals. It can be seen from this table that the period trend in atomic radii is not followed as closely by the transition metals. While the elements on the right side of the periodic table are non-metals. The elements found on the left side of the periodic table are typically metals. Elements can be categorized into three major groups that include metals, nonmetals, and metalloids. He discovered, for example, that the atomic masses previously reported for beryllium, indium, and uranium were incorrect. Table above lists the atomic radii for the first row of the transition metals. The atomic number refers to the number of protons found in the atom of an element. When the chemical properties of an element suggested that it might have been assigned the wrong place in earlier tables, Mendeleev carefully reexamined its atomic mass. 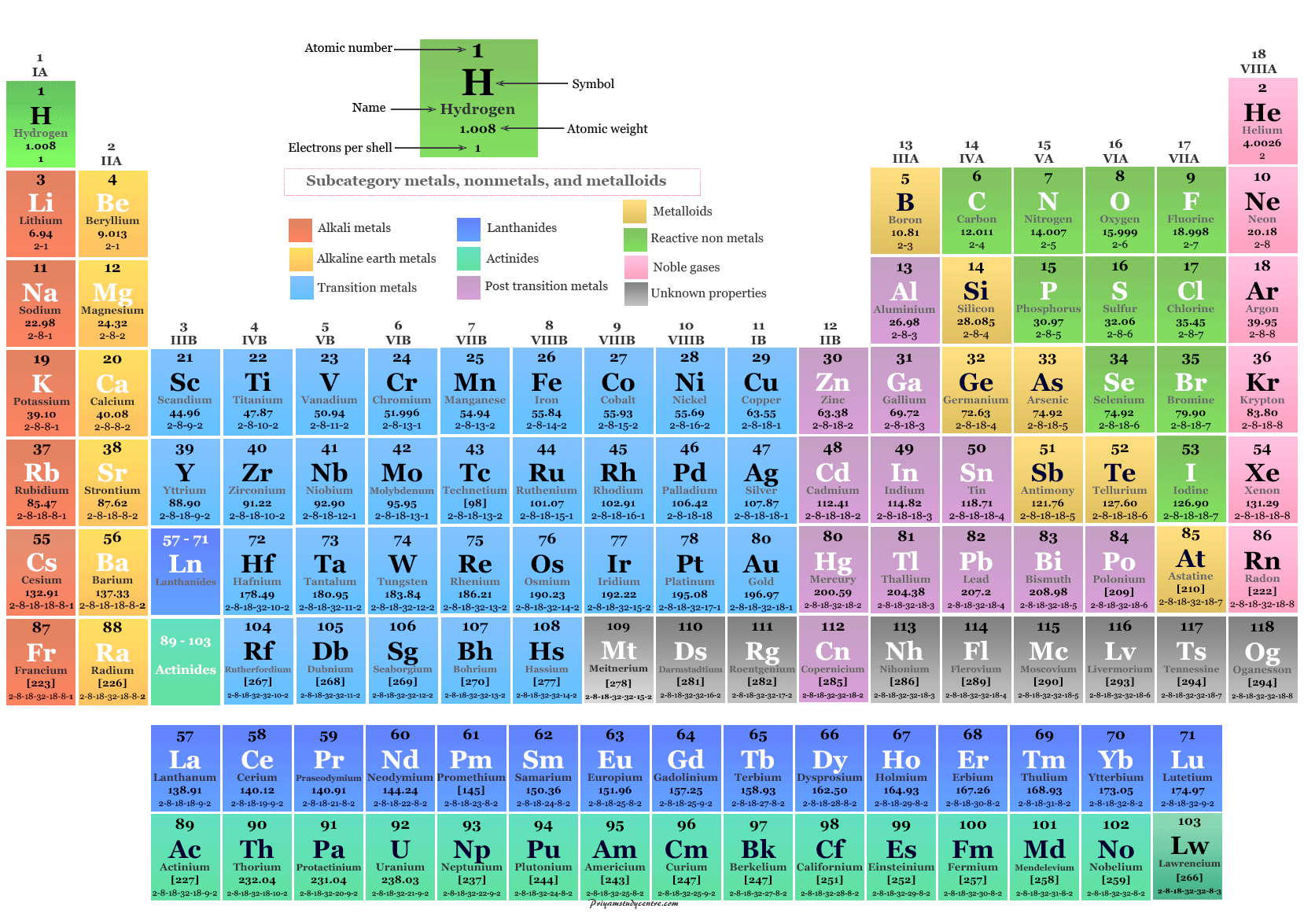
These are the first 20 elements, listed in order: H - Hydrogen. This is also the number of protons in each atom. \): Comparison of the Properties Predicted by Mendeleev in 1869 for eka-Aluminum and eka-Silicon with the Properties of Gallium (discovered in 1875) and Germanium (discovered in 1886). The elements are ordered in the periodic table according to increasing atomic number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
